In physics, no measurement is ever 100% exact. Understanding the difference between how "correct" a measurement is versus how "consistent" it is represents the core of experimental science.
1. Accuracy vs. Precision
These two terms are often used interchangeably in daily life, but they have very different meanings in a laboratory setting:
- Accuracy: How close a measured value is to the true or accepted value.
- Precision: How close a series of measurements are to each other (repeatability).
The Dartboard Analogy
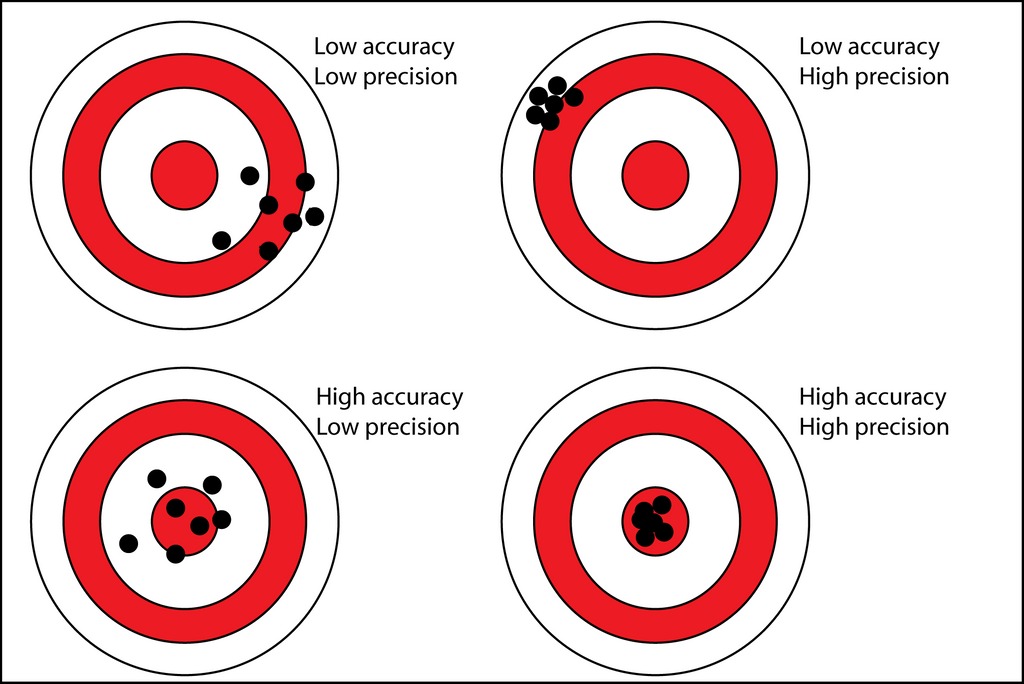
- High Accuracy, Low Precision: Darts are scattered around the bullseye.
- Low Accuracy, High Precision: Darts are all bunched together, but far from the bullseye.
- High Accuracy, High Precision: All darts are in the bullseye.
2. Types of Experimental Errors
Errors are not "mistakes"; they are uncertainties inherent in the measurement process.
A. Systematic Errors
These errors shift all measurements in the same direction. They affect Accuracy.
Example: A weighing scale that isn't "zeroed" properly (Zero Error) or a ruler that has shrunk due to heat.
Fix: Calibrate the instruments or use a correction factor.
B. Random Errors
These are unpredictable fluctuations that affect Precision.
Example: Slight variations in reaction time when using a stopwatch or air currents affecting a sensitive balance.
Fix: Take multiple readings and calculate the mean (average).
3. Quantifying Uncertainty
To communicate how reliable a measurement is, we use specific formulas.
Absolute Error ($\Delta x$)
The physical difference between the measured value ($x_m$) and the true value ($x_t$).
$\Delta x = |x_m - x_t|$
Relative Error
The ratio of the absolute error to the true value.
$\text{Relative Error} = \frac{\Delta x}{x_t}$
Percentage Error
The relative error expressed as a percentage. This is the most common way to report error in IJSO practicals.
$\text{Percentage Error} = \frac{|x_m - x_t|}{x_t} \times 100\%$
4. Significant Figures (Sig Figs)
Significant figures tell the reader how precise your measuring tool was.
- Rule 1: All non-zero digits are significant (e.g., 1.25 has 3).
- Rule 2: Zeros between non-zero digits are significant (e.g., 105 has 3).
- Rule 3: Leading zeros are never significant (e.g., 0.002 has 1).
- Rule 4: Trailing zeros in a decimal are significant (e.g., 2.50 has 3).
💡 Lab Pro-Tips
- Parallax Error: Always look at a scale (like a graduated cylinder or ruler) directly at eye level. Looking from an angle introduces a systematic error.
- The Power of Averaging: If you have five different results for the same experiment, the average is almost always more accurate than any single result.
Finished studying this topic?
Mark it as completed to unlock the next level in your Physics journey!